Covid-19 vaccine: Who will win the race?
The race for a vaccine for Covid-19 has begun, with the US and China in the lead with clinical trials and testing. Oxford University visiting researcher Hayson Wang points out that countries will have to work together in order to develop an effective vaccine, rather than compete against one another.
As Covid-19 sweeps the globe, many are focusing their attention on developing a vaccine as a way to control the spread of the virus that has no effective treatment to date.
The difficulties in vaccine research
A successful vaccine has to satisfy many criteria such as effectiveness, safety and timeliness. Moreover, it has to be tested on animals and humans before being put on the market.
First, effectiveness. Covid-19, like the HIV and Ebola viruses, are RNA (ribonucleic acid) viruses. These viruses have a much faster mutation rate compared to the DNA (deoxyribonucleic acid) viruses. As RNA viruses undergo rapid evolution, it is difficult to develop an effective vaccine. A vaccine for the HIV virus has been under development for over 30 years, but even with over 40 vaccines being developed globally, not a single one has made it to clinical testing, because none has elicited an effective human immune response.
And due to insufficient initial research into the genetics, immunology, and epidemiology of Covid-19, it is very difficult to develop a vaccine. Trying to find a quick breakthrough without a solid early foundation adds to the likelihood of taking a roundabout route and wasting time, or worse, leading to a less effective vaccine and failed research.
Currently, eight vaccine candidates are in clinical trials - four from China, two from the US, one from the UK and one from Germany working in collaboration with an American and a Chinese company.
Second, safety. Vaccines must not harm the human body after administration, and adverse effects have to be kept to a minimum. This involves a series of complicated processes and testing on animals and humans, so there is a certain insurmountable timeline to developing vaccines. Even with government support in terms of funding, manpower, and policies, the laws of research cannot be overcome.
Progress in China and the world
As of late April, over 100 research projects on a vaccine for Covid-19 have been announced worldwide, with clear progress on 78 projects. Most of these (46%) are in North America, with 14 in China. Currently, eight vaccine candidates are in clinical trials - four from China, two from the US, one from the UK and one from Germany working in collaboration with an American and a Chinese company.
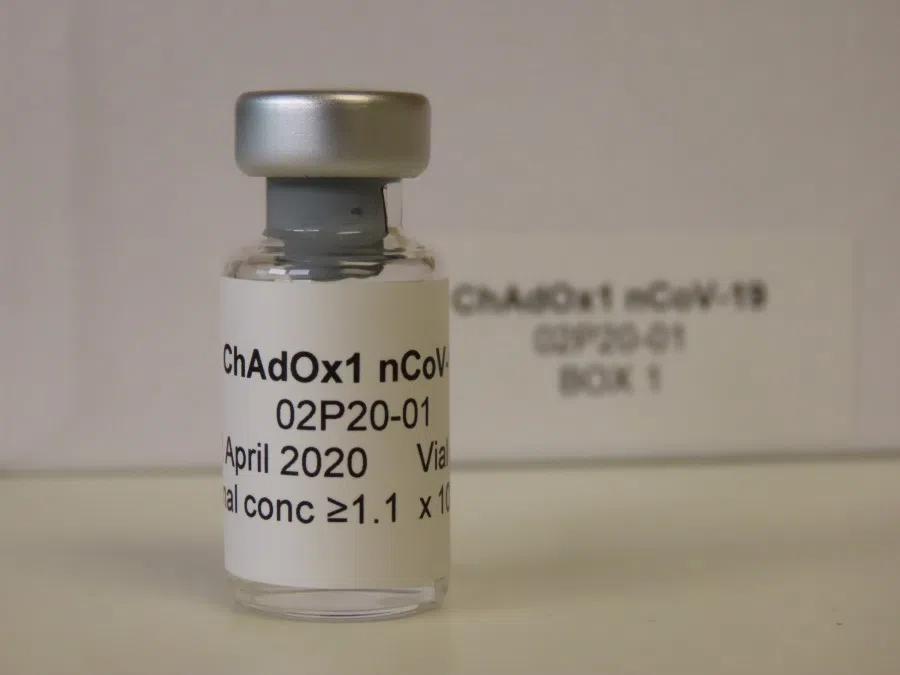
Of these efforts, Oxford University's adenovirus vector-based vaccine (made by a genetic restructuring technique) has been tested on over 500 people in phase one trials, with the possibility of expanding it to thousands of people, making it the first vaccine to enter large-scale clinical trials. (NB: A total of about 1,100 people were involved in the trial, the other half being a control group receiving a meningitis vaccine. There are plans to conduct a 5,000-person trial by June.) Another adenovirus vector-based vaccine by the PLA Academy of Military Science, has gone into phase two of clinical trials, the fastest to do so. Another inactivated vaccine (one that uses the killed version of the virus that causes a disease) from the Wuhan Institute of Biological Products, has also gone into phase two of clinical trials.
However, adenoviral vectors themselves can also elicit an immune response in the host, and their effect on the human immune system is complex.
Inactivated vaccines are made by making a virus non-infectious while retaining its ability to elicit an immune response. The development process for inactivated vaccines is straightforward, but their protective effects are not long-lasting, and repeated doses have to be administered for sustained immunity, while carrying a higher risk of adverse reactions.
Adenovirus vector-based vaccines are made by a genetic restructuring technique where harmless segments of coronavirus genes are inserted into an inactivated adenoviral vector. When injected, the adenoviral vector will infect human cells and deliver the coronavirus genes that it is carrying, prompting the immune system to produce antibodies. However, adenoviral vectors themselves can also elicit an immune response in the host, and their effect on the human immune system is complex.
From the current literature, the team led by Professor Qin Chuan of the Institute of Laboratory Animal Sciences under the Chinese Academy of Medical Sciences and Peking Union Medical College has administered an inactive vaccine candidate for Covid-19 into rhesus macaques, and this has been shown to be safe. This is the first publicly reported result of a Covid-19 vaccine being clinically tested on animals. However, there is currently no clinical test data, and it will take time to verify the effectiveness and safety of various vaccines. (NB: Most recently, US company Moderna helming one of the eight clinical trials under way, has reported promising first results. Eight people who received two doses of the experimental vaccine starting in March were found to have matching or higher levels of neutralising antibodies than in recovered Covid-19 patients.)
Politicisation of science
The importance of a vaccine is obvious - there is currently no effective treatment for the coronavirus, stay-at-home and lockdown measures are creating a huge economic impact. Vaccine research and development has become a scientific battlefield, and even the main arena for competition among countries. Governments are putting in funds and manpower, and encouraging scientific research organisations and R&D companies to join in.
At the same time, the coronavirus has brought about changes in international affairs. Some Western countries, led by the US, are in a war of words with China, and the development of a vaccine would be to some extent an important asset for China in future international competition.

However, with everyone busy fighting the coronavirus, the US government has been engaging in some political manoeuvring. It has been revealed that the US has tried to get a German vaccine company to move its research to the US, while the FBI and the Department of Homeland Security issued a public service announcement saying that "PRC-affiliated cyber actors and non-traditional collectors" have been observed "attempting to identify and illicitly obtain valuable intellectual property (IP) and public health data related to vaccines, treatments, and testing from networks and personnel affiliated with Covid-19-related research".
The world is hoping for a vaccine for the coronavirus, which calls for countries to work together. If this can be quickly accomplished through sharing of information and international cooperation, that would be a most impressionable miracle for the history books.
"COVID-19 vaccine development and deployment in China, when available, will be made a global public good. This will be China's contribution to ensuring vaccine accessibility and affordability in developing countries." - Chinese President Xi Jinping
Note:
Even while the vaccine is being developed, the question of how any future vaccine will be distributed is already creating tensions among countries.
Following an announcement by French pharmaceutical company Sanofi that it would give the US priority to a coronavirus vaccine, French Prime Minister Édouard Philippe gave a public apology on 14 May, saying that such a vaccine would be given to all countries as a public good.
US Secretary of State Mike Pompeo has also said that he hopes that any successful vaccine will be quickly and effectively distributed throughout the world. Reuters quoted Pompeo telling Israel's news agency Kan 11 on 13 May, "I hope that we will all collectively find a way to produce this at high volume to get it all across the world and make sure that every citizen that needs access to a vaccine can get it as quickly as possible."
At the opening of the World Health Assembly on 18 May, Chinese President Xi Jinping said, "COVID-19 vaccine development and deployment in China, when available, will be made a global public good. This will be China's contribution to ensuring vaccine accessibility and affordability in developing countries."

![Hokkien roots, Brunei soil: My father’s journey across borders and spiritual planes [Eye on Fujian series]](https://cassette.sphdigital.com.sg/image/thinkchina/71f359b221f9d122e33e939974a2a8159629f5990340bdf70135adfea2d6a086)
![[Video] Jereme Leung: From Nanyang to Jiangnan — where taste becomes memory](https://cassette.sphdigital.com.sg/image/thinkchina/915587e6ef38f403df0792bdbbf90b20fc4fb5d39b299feacefd74b9bf8e685d)


